 If the result from amplified DNA from the embryo biopsy falls in the mosaic range then the embryo is now classified as mosaic and potentially abnormal. This can be translated to the results from biopsies on real embryos. The relative change in the copy number calls can be plotted out for different levels of mosaicism. Mixtures of normal and abnormal cells are created and then the DNA is amplified and analyzed in a similar fashion to PGS. Several studies on the detection of mosaicism have focused on first conducting experiments with different cell lines in the laboratory. What if there are nests of abnormal cells that fall outside the tiny TE biopsy? Even if the biopsy captures a mix of two different cell types, will our technologies accurately be able to detect that fact? First of all, some mosaicism may not be captured by a single biopsy. 
A much greater challenge is to determine if there is a mixture of normal and abnormal cells within a single biopsy, indicating mosaicism. Several studies have shown that PGS can be accurately performed with microarrays, real-time polymerase chain reaction and next generation sequencing to select euploid embryos with a higher chance of implanting. This amplification step and secondary analysis has the potential to introduce errors. Rather, the aggregate of the DNA from the biopsy is extracted from the cells and amplified (copied) millions of times over to be able to obtain a quantity sufficient for analysis. The DNA also cannot be directly analyzed from individual cells. But the amount of DNA obtained in a TE biopsy for PGS is miniscule, just several picograms worth, and that creates a problem because the cells cannot be cultured or visually inspected. Some cells may have the normal complement of 46 chromosomes (22 pairs of autosomes numbered 1-22 and the sex chromosomes X and/or Y), and some cells may have 45 (due to monosomy or 1 copy of a given chromosome) or 47 chromosomes (due to trisomy or 3 copies of a given chromosome). Those latter tests involve actually culturing cells and visually inspecting the structure of the chromosomes in dozens of cells. Diagnosing mosaicism in a TE biopsy is very different from diagnosing it in an amniocentesis or blood test. A separate collection of cells called the inner cell mass (ICM) becomes the actual embryo, and ultimately the baby, and is not touched by the embryologist when TE biopsy is performed. 
A study performed at Reproductive Medicine Associates of New Jersey demonstrated that TE biopsy is safer than biopsying a single cell on day 3, and it’s also more accurate. This outer layer, known as the trophectoderm (TE), eventually becomes the placenta and membranes. The PGS Diagnostic ProcessĪs currently performed, PGS involves using a laser to biopsy several cells (typically 5-10) from the outer layer of a blastocyst. Depending on the chromosome involved, and the proportion of abnormal cells, mosaicism could lead to embryos that cannot implant, that miscarry or that result in pregnancies with abnormalities (mosaicism confined to the placenta has been associated with abnormal growth of the fetus). Simply stated mosaicism is when an embryo, or person, is composed of two or more genetically different cell types. But before considering the challenges of mosaicism and what to do with predicted mosaic embryos, it is important to take a step back and carefully consider what we know, and what we don’t know, about mosaicism. This has resulted in a shift in how we classify PGS-tested embryos: from the previous “normal or abnormal” dichotomy, to now a spectrum that includes a more ambiguous, “mosaic” designation. However, as genetic technologies with higher resolution have been applied to PGS, the ability to reliably detect more types of genetic errors, such as mosaicism, has improved. It’s important to realize that nothing has changed intrinsically about the embryos transferred in IVF for decades we have been transferring unscreened embryos that included a mix of chromosomally normal (euploid), chromosomally abnormal (aneuploid) and mixed (mosaic) embryos. 
High-profile articles have in the New York Times and the New England Journal of Medicine, focusing on the challenge of what to do with mosaic embryos created during IVF. This latest one revolves around the issue of mosaicism or embryos that predicted to be a mix of normal and abnormal. 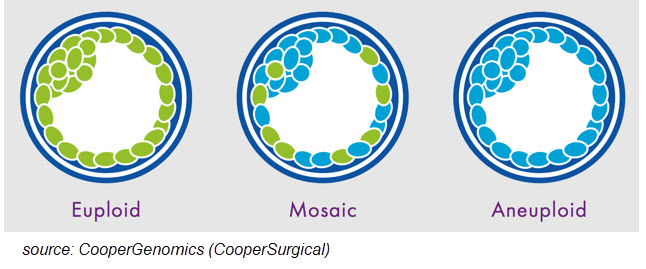
But just when we thought the controversies surrounding PGS had been mostly resolved, over the last several months a new controversy has emerged. It is now routine to transfer a single embryo and maintain excellent pregnancy rates and minimize the risk of miscarriage across age groups. The increased use of preimplantation genetic screening (PGS) in recent years has resulted in improved outcomes for couples with infertility who are going through IVF. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
